Clarametyx Biosciences Announces Progress on Study of Antibody Therapy CMTX-101 For Infections Associated With Cystic Fibrosis
Following successful Phase 1b completion, Clarametyx is initiating the Phase 2a portion to assess CMTX-101 as an adjunct to standard of care for chronic infections
COLUMBUS, Ohio – October 17, 2024 – Clarametyx Biosciences Inc. (“Clarametyx”), a biotechnology company developing targeted, immune-enabling biologic therapies to counter serious infections associated with biofilms, today announced that it has completed the initial Phase 1b portion of its clinical trial evaluating the novel immune-enabling antibody therapy CMTX-101 to treat cystic fibrosis (CF)-associated pulmonary infections and is now initiating the Phase 2a portion of the trial.
“The CF population is vulnerable to recurrent and chronic lung infections, resulting in the need for interventions that can enable more effective clearance of the bacteria to reduce courses of antibiotics,” said Jerry Nick, MD, Professor in the Division of Pulmonary, Critical Care and Sleep Medicine at the National Jewish Health Department of Medicine, and primary investigator of the study. “We are encouraged by the initial data from the 1b portion of the study, which demonstrated no safety signals with CMTX-101. If clinically confirmed to disrupt bacterial defenses and improve response to antibiotics and natural immunity, CMTX-101 will add a new treatment strategy for people with CF.”
This double-blind, randomized, placebo-controlled trial is designed to assess the safety and tolerability, PK, immunogenicity, reduction of pulmonary P. aeruginosa burden, and exploratory endpoints of CMTX-101 as an adjunct therapy to standard of care antibiotics in people with CF. Following the completion of the Phase 1b period and a review of safety data and recommendation by independent safety review committees, the Phase 2a study portion now aims to enroll up to 41 adults diagnosed with CF to evaluate the safety and tolerability of CMTX-101. The company expects to report initial findings from the study in 2025. More information on the study and participating sites is available at ClinicalTrials.gov (http://www.clinicaltrials.gov/) using the identifier NCT06159725.
The advancement of the technology in CF-associated infections is aligned with the company’s strategic focus on addressing recalcitrant and chronic infections caused by a range of bacterial pathogens. The company recently concluded an early-stage clinical study of CMTX-101 in community-acquired bacterial pneumonia, generating valuable safety, tolerability, pharmacokinetic and immunogenicity data, along with exploratory efficacy insights. Importantly, the study found no major safety signals, minimal anti-drug antibodies and no neutralizing antibodies. Clarametyx is presenting positive topline findings from the study on October 18th at IDWeek in Los Angeles.
“We recognize that recurrent infections are a significant challenge for people living with cystic fibrosis and their families, and believe that a truly novel approach to help the patient’s immune system and today’s antibiotics more effectively clear these infections can not only alleviate the burden on these patients but also support greater antibiotic stewardship and potentially reduce resistance issues,” said David Richards, Chief Executive Officer. “With encouraging early data from the Phase 1b portion of our CF study, we are eager to advance into the next phase of development to demonstrate the strong potential of our technology to redefine treatment paradigms for chronic and persistent infections.
About CMTX-101
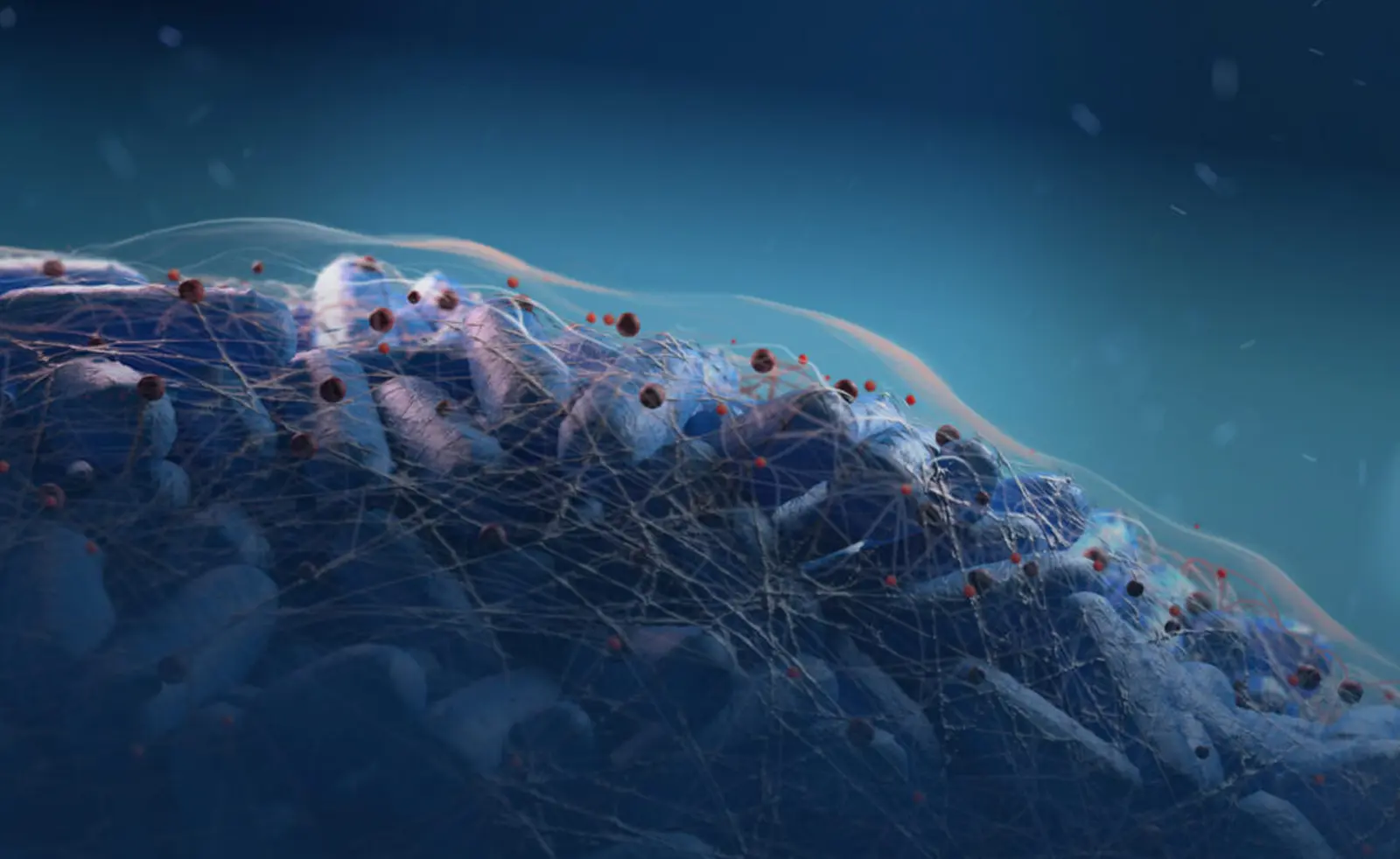
CMTX-101 is an immune-enabling antibody therapy designed to precisely and rapidly destroy the universal underlying structure of bacterial biofilms to undermine extracellular bacterial defenses and enable more effective antibiotic and immune intervention. Because the target is universally present across bacteria, the technology can be employed to treat a range of bacterial infections, many of which are characterized by the presence of multiple types of bacteria. CMTX-101 is intended to be administered concomitantly with a wide range of standard-of-care antibiotics that target both Gram-negative and Gram-positive bacteria. The goal of treatment is to dramatically improve the effectiveness of antibiotic therapies and innate immune system effectors, improving the time to resolution of the infection and reducing the need for repeated courses of antibiotics.
About Clarametyx Biosciences
Clarametyx Biosciences is combating the formidable challenge of persistent and recalcitrant infections through an innovative technology platform targeting the biofilm—a protective layer around bacteria—to enable a more effective immune response and antibiotic intervention. The Columbus, Ohio-based company is building a pipeline of immune-enabling therapies and vaccines for life-threatening bacterial infections associated with biofilms, with a near-term focus on challenging respiratory infections. For more information, visit us on the web or on LinkedIn.
Media Contact:
Kellie Hotz
khotz@clarametyx.com
# # #